REAL RISKS of COVID VACCINATION
**This page was last updated on January 4, 2023 and will not be updated going further.**
“In countries where the vaccine has not been approved by the relevant regulatory authority, it is an investigational drug, and its safety
and efficacy have not been established.”
– “Global Information About Pfizer-BioNTech COVID-19 Vaccine (also known
as BNT162b2)," Pfizer, accessed June 27, 2021, https://www.cvdvaccine.com
BELOW:
______________________________________________________
REAL RISKS of COVID-19
VACCINATION
Make an Informed Choice!
More than 660 million doses of COVID-19 vaccines were administered in the U.S. from December 14, 2020, through December 14, 2022, according to the CDC on December 21, 2022. Although long-term effects are as yet unknown, many of the vaccinated have experienced little to no difficulty. However, many other Americans have experienced mild to severe adverse events, which is not often mentioned in news reports pushing vaccination. The pro-vaccine, vaccine-injured individuals speaking at U.S. Senator Ron Johnson's June 28, 2021 press conference on adverse reactions want to be seen, heard, and believed.
- Sheryl Ruettgers (wife of Ken Ruettgers, former Green Bay Packer): "I am pro-vaccine. I am definitely pro-science, but I'm also pro-informed consent. And these vaccine injuries are real, but people don't know."
- Kristi Dobbs: "I would give anything; I would give my whole life savings if I could go back to January 17th and never have had this experience in my life."
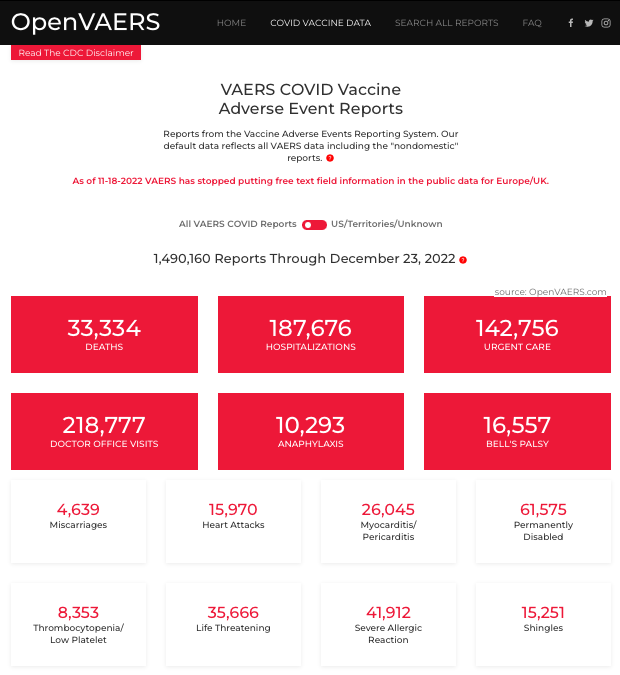
As of December 23, 2022, OpenVAERS includes 1,490,160 reports of adverse events due to the covid injection, which can include everything from death to simply getting the wrong dose. Find a screenshot of one report below. If you are contemplating vaccination for COVID-19, take this opportunity be more fully informed and then make your own decision. Here are several considerations and some of the significant adverse reactions that have been reported after vaccination:
-
Death – 18,007 deaths reported after vaccination to VAERS per the CDC, as of December 21, 2022.** The CDC "has identified nine deaths causally associated with the J&J/Janssen COVID-19 vaccination." Specifically, CDC says VAERS reports "do not necessarily mean that a vaccine caused a health problem." However, since information in VAERS reports is often "incomplete, inaccurate, coincidental and unvarifiable," a determination of causation may be difficult. To investigate adverse events, the CDC will often request medical records. To make it easier to see inside the complex VAERS system, OpenVAERS was created by an independent group. It lists 33,334 deaths reported as of December 23, 2022.
-
Anaphylaxis and allergic reactions
-
Vascular: Thrombosis (blood clots), nose bleeds, stroke, emboli, thrombocytopenia (low platelets)
-
Cardiac and Respiratory: cardiac arrest, cardiac arrhythmia, atrial fibrillation, shortness of breath, pulmonary emboli
-
Inflammation: lymph node swelling; 26,045 reports to VAERS of myocarditis/pericarditis (especially high in young men)
-
Nervous system disorders: stroke, Guillain-Barre, Bell’s palsy, seizure, temporary paralysis, unable to walk
-
Muscle spasms and twitching – for weeks or months
-
Pain – extremities, chest, kidney, abdominal, eye, “pins and needles,” injection site
-
Pregnancy adverse events – spontaneous abortion, miscarriage, stillbirths, menstrual changes, maternal and fetal death, and possible infertility problems
-
Sensory/Cognition – headache, loss of sight, hearing, speech, tinnitus, brain fog, cognitive decline
-
Compromised immune system
- Abdominal: diarrhea, colitis, vomiting, gastrointestinal bleeds
- Shingles; rashes, swelling
-
Antibody-Dependent Enhancement (ADE) – more severe COVID illness after the vaccinated are exposed to live virus
-
SIRVA – Shoulder Injury Related to Vaccination Administration
-
Extreme fatigue; fever; fainting, dizzy spells
-
"Breakthrough" COVID-19 infection – On July 30, 2021, NBC reported 125,682 cases in 38 states but says the numbers are likely higher because of the non-reporting states; CDC had reported 3,016 hospitalized or fatal breakthrough cases, as of June 1, 2021; on May 1, 2021, the CDC stopped collecting data on breakthrough cases that did not lead to hospitalization or death, despite 10,000 breakthrough cases reported through April 30, 2021. Therefore, the actual number is unknown. NOTE: "breakthrough" is the wrong word for these infections. On July 20, Dr. Fauci told the U.S. Senate that the vaccine's efficacy was based on preventing "clinically apparent disease" (symptoms), "not preventing infection."
-
Little to no benefit from vaccination if already had COVID, according to a Cleveland Clinic study which states: “Individuals who have had SARS-CoV-2 infection are unlikely to benefit from COVID-19 vaccination, and vaccines can be safely prioritized to those who have not been infected before."
-
Also, some counties with higher vaccination rates have higher COVID-19 case rates, while case rates are falling in some counties with below average vaccination rates (CBS Sacramento, July 26, 2021)
-
Unknown clinical implications of COVID-19 spike protein and S1 protein circulating in blood post-vaccination.
-
Unknown long-term effects – because the vaccine was rushed under emergency use authorization (EUA)—"Clinical trials alone take six to seven years on average to complete"—long-term effects are unknown.
This is not an all-inclusive list.
Click here for all sources and citations for above data
______________________________________________________
Reporting Systems
The federal Vaccine Adverse Events Reporting System (VAERS) is not user-friendly. Created by Congressional mandate in 1990, it is difficult both to report data and to view the reported data. A reporter of an adverse event, which can be anyone (vaccine injured person, parent, friend, physician, health care worker, hospital administrative staff, health plan, etc.), must complete approximately five to six pages of information for each individual report. Here are the 10 things the CDC wants physicians and other health care practitioners to know about VAERS reporting for COVID-19, and CDC use of data for investigations. Read also the CDC's description of VAERS.
Notably, the FDA requires "Vaccination providers enrolled in the federal COVID-19 Vaccination Program must report all vaccine administration errors, all serious adverse events, cases of Multisystem Inflammatory Syndrome (MIS) in adults and children, and cases of COVID-19 that result in hospitalization or death following administration of" the vaccine. The CDC includes recommended reporting, which can include any clinically significant event post-vaccination, "even if they think the event might not be related to the vaccine," and required reporting.
The reported case is given a temporary VAERS ID number. After the CDC has reviewed the case (and possibly requested supporting data and records from the reporter), the case may be given a permanent VAERS ID number. If so, that case is added to, and can be found in, the VAERS. The temporary ID is not included in the report in VAERS so it is difficult for adverse event reporters to know if their case is actually included on the VAERS. The number of reported cases not included on the VAERS appears to be unknown.
NOTE: VAERS warns adverse-event reporters that they are subject to fines and imprisonment if they falsely report a case.
It is also difficult to accurately view the data in VAERS. According to several sources, the data is reported by year, but in three separate reports. Those who wish to properly understand the data must ask for the correct information, using the correct fields (including, according to OpenVAERS, requesting only the "received date"—the date the report was received by VAERS). There is no data field for the date the vaccine was given, although it could be written in the narrative. The three data sets must then be pulled and combined.
Thus, OpenVAERS was created about a year before COVID-19 by a small group of individuals (who have personally experienced vaccine injury) to make it easier for the general public to see the injury and adverse event data available on VAERS. OpenVAERS pulls data directly from VAERS without manipulation: "We do not change, modify or vet data. We take the downloads, upload them to our server and put a different face on them so they are easier to browse and get accurate information from."
Screenshot of an Individual Report Viewed in OpenVAERS:
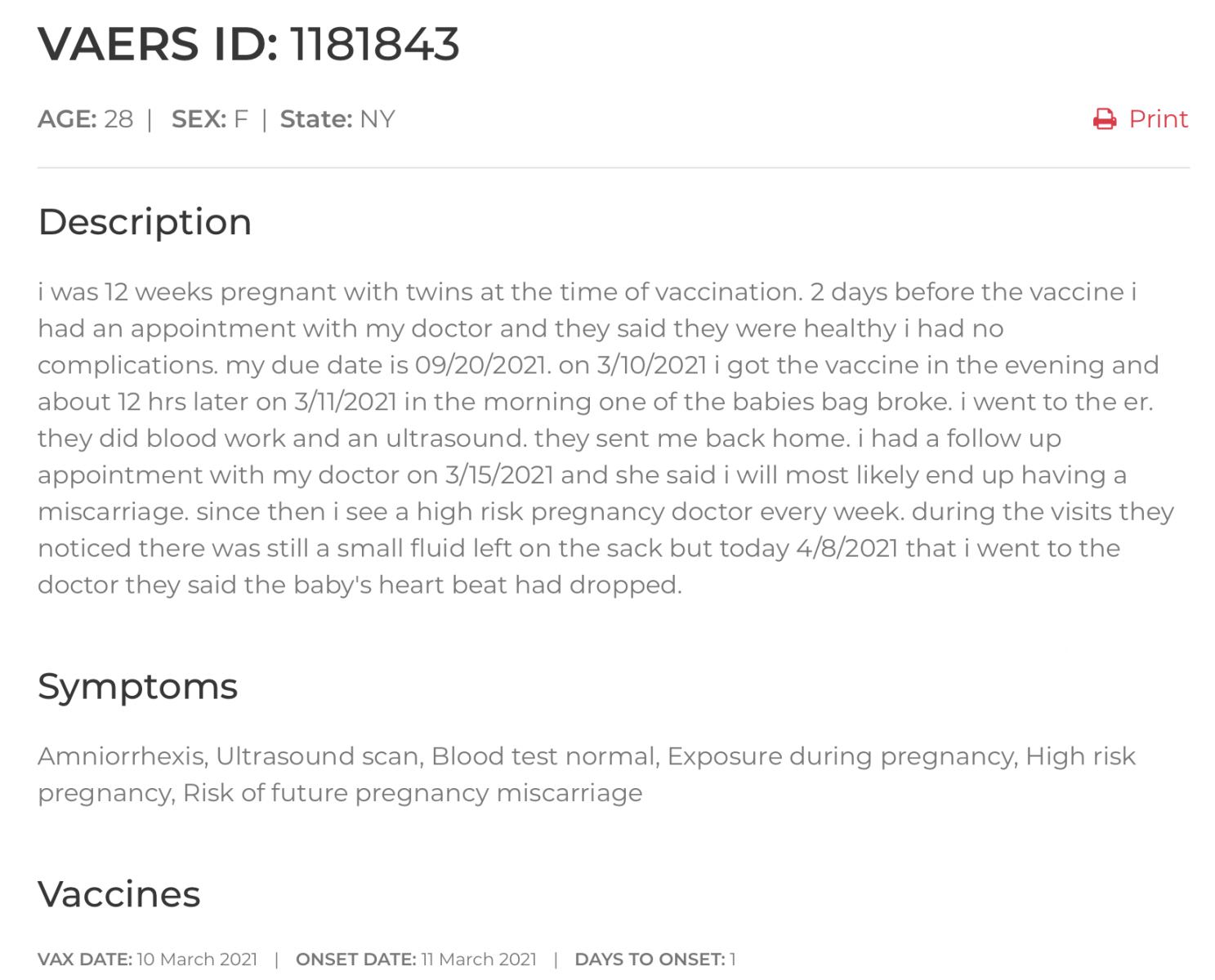
Screenshot from OpenVAERS: (click for latest update)
______________________________________________________
Print this document and share it with your employer, school, etc. if it
attempts to mandate the Covid-19 "vaccine." (See CDC definition in citations)
.jpg)
In an official document found by David Martin, Ph.D., an expert in patents and other
intangible assets, and included in his publication, "The Fauci/COVID-19 Dossier,"
the U.S. Patent Office once denied a patent to Dr. Fauci for a HIV vaccine
because it did not "prevent infection":

______________________________________________________
SAYING NO
While some are encouraging President Biden to withhold federal dollars from states like Florida that prohibit vaccination mandates (11 so far), the following Americans have a variety of reasons for refusing to take the injection regardless of what that means for employment, education or the risk of infection:
-
"Christian quarterback resists NFL's vaccine pressure," LifeSite, August 31, 2021
-
"Why so many tennis players don't want the Covid vaccine," Ben Rothenberg, New York Times, August 30, 2021
-
"153 people resigned or were fired from a Texas hospital system after refusing to get vaccinated," Dan Diamond, The Washington Post, June 22, 2021.
-
"Broadway star and MN native Laura Osnes says she wasn't fired, she quit gig after refusing vaccine," Ross Raihala, Pioneer Press, August 17, 2021.
-
"Offspring drummer says band axed him from tour for not getting vaccinated," Lisa Respers France, CNN, August 4, 2021.
-
"Unvaccinated man hospitalized with COVID-19 still refuses to get the vaccine," Lee Moran, HuffPost, July 23, 2021.
-
"Bills WR Cole Beasely: I'm not anti-vaccine, I'm pro-choice," Alex Braskey, Livingston County News, July 28. ("For so many players around the NFL, safety does not solely mean avoiding the COVID virus. Our health is the now and years beyond that we are trying to protect with our personal choice along with doing all the things we did in our protocol during a very successful 2020 NFL season.")
-
"Many Amish in America unsure about Vaccine," John Russell, VOA Learning English, July 4, 2021
-
"She's leading the fight against mandatory vaccines in Texas. She also happens to be a nurse." Peter Holley, Texas Monthly, May 19, 2021.
-
"Don't call us names for refusing the vaccine," (Letter to the Editor), Greg Groh, Houston Chronicle, March 29, 2021
-
"Nearly 40% of Marines decline COVID-19 vaccine, prompting some Democrats to urge Biden to set mandate for military," Sarah Elbeshbishi, USA TODAY, April 11, 2021
______________________________________________________
CCHF has created a specific page of resources to use in conversations with employer and/or school or to request legal assistance, including forms:
______________________________________________________
with only 35 hospitalized and 14 deaths (all with diabetes).
DON'T DELAY!
IMPORTANT STUDY
"Review of Emerging Evidence Demonstrating the Efficacy of Ivermection in the Prophylaxis and Treatment of COVID-19" Pierre Kory, MD, et al, American Journal of Therapeutics, May/June 2021
"In summary, based on the totality of the trials and epidemiologic evid3nce presented in theis review along with the preliminary findings of the Unitaid/WHO meta-analysis of treatment RCTs and the guideline recommendation from the international BIRD conference, ivermectin should be globally and systematically deployed in the prevention and treatment of COVID-19." [emphasis ours]
LONG HAUL COVID
In his June 24, 2021 interview on DrBeen Medical Lectures, Dr. Bruce Patterson, MD, co-founder of the Chronic COVID Treatment Center (which has a network of 100 physicians nationwide), says patients with "Long Covid" are not only those who were infected with Covid, but the vaccinated who have never had Covid—due to the Covid spike protein: "Spike Proteins In Immune Cells - Dr. Bruce Patterson Discusses COVID Long Haul." Dr. Mobeen restating an earlier guest said, "Long haul does not occur if you're treating with ivermectin" (21:00 mark)
______________________________________________________
Early SARS-CoV-2 did not appear to be easily transmitted, as people in households experienced. Published in February 2021, this Imperial College London study of transmissibility between 'close contacts' found the following transmission chances:
-
3.5% if the person infected with COVID-19 had no symptoms
-
12.8% if the infected person had symptoms
-
21.1% if the infected person lived in the same household.
In short, if the infected person lived in the house, household members have a nearly 80% chance of not getting COVID-19. See screenshot below.
-
"COVID-19 spread in diffrent social settings - new Imperial report," Dr. Sabine L. van Elsland, Imperial College London, November 27, 2020: https://www.imperial.ac.uk/news/209673/covid-19-spread-different-social-settings-imperial/
-
"Severe Acute Repiratory Syndrome Coronavirus 2 (SARS-CoV-2) Setting-specific Transmission Rates: A Systematic Review and Meta-analysis," Hayley A Thompson, et al, Clinical Infectious Disease, February 9, 2021: https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciab100/6131730
Screenshot from Imperial College report:

Regarding the Delta variant, the transmission is said to be higher but whether it is more virulent is in question (all emphasis ours below):
-
Yale Medicine (July 30, 2021) reports: "One important question is whether the Delta strain will make you sicker than the original virus. Early information about the severity of Delta included a study from Scotland that showed the Delta variant was about twice as likely as Alpha to result in hospitalization in unvaccinated individuals, but other data has shown no significant difference. The information could change as experts learn more."
-
In "Why No One Is Sure If Delta Is Deadlier," an June 28, 2021 article in The Atlantic, the reporter talked with Paul Turner, an evoluntionary biologist and virologist at Yale and reported this from their conversation: "no evidence so far suggests that the coronavirus is systmatically evolving to become more malicious."
CASES — COVID-19 in USA (as of July 30, 2021)
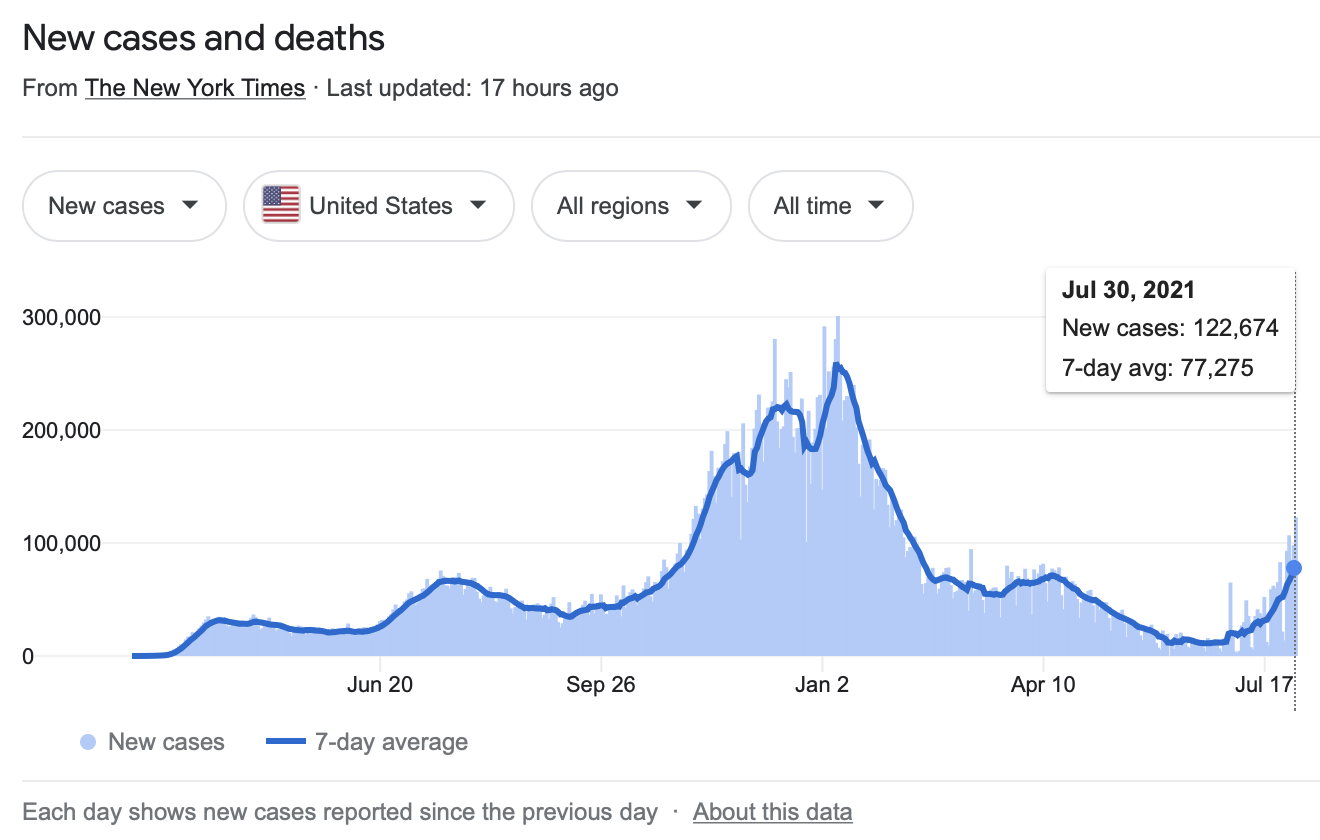
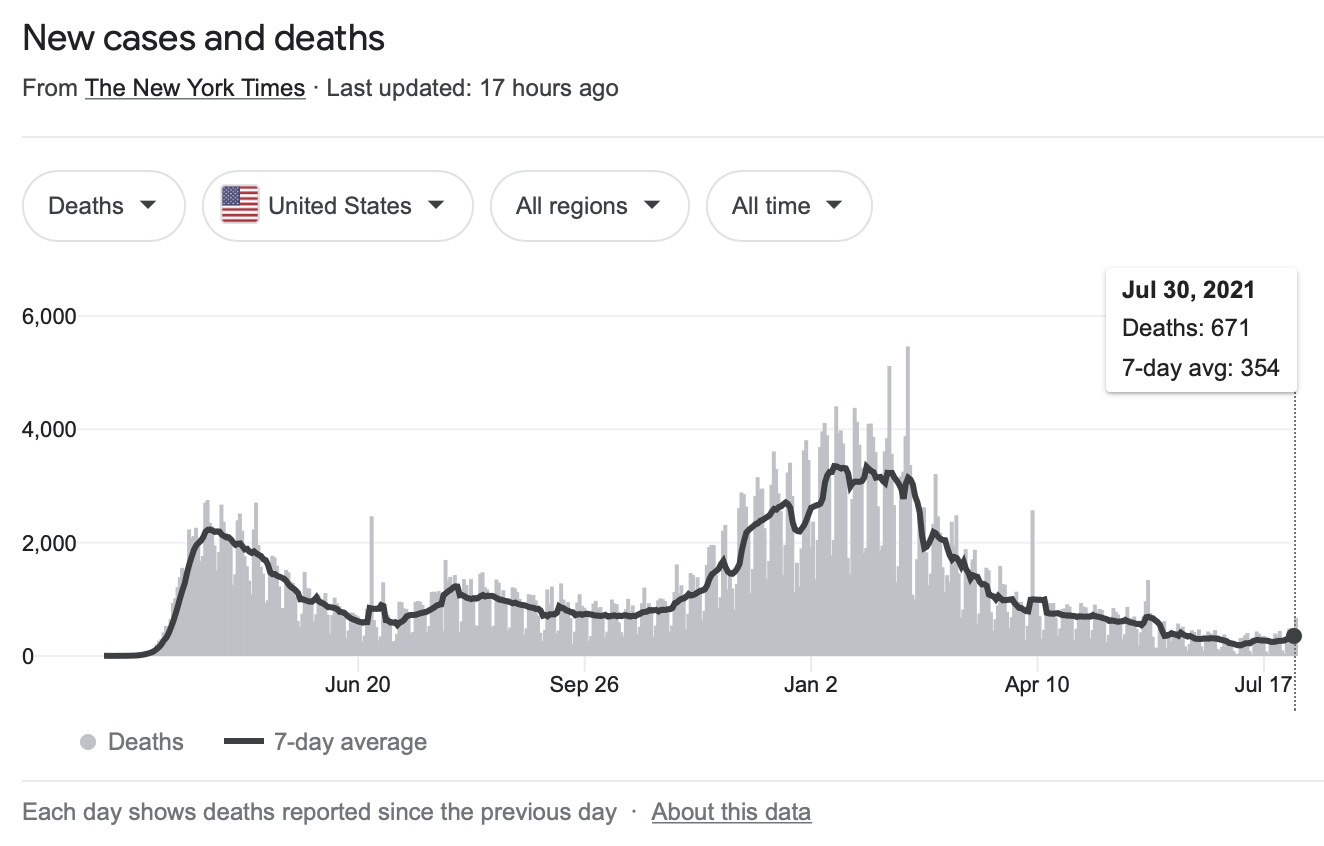
DEATHS — COVID-19 in USA (as of July 30, 2021)
** The CCHF billboard includes the number of reported deaths published by the CDC as of July 19, 2021 (a plus sign was used to designate the increase in reports sure to come).







